Bringing a neuromodulation medical device to clinical reality is rarely just engineering. It is a long, tightly interdependent chain of decisions: electrode design and materials, implantable electronics and embedded software, system integration, biocompatibility testing, and regulatory documentation, each one affecting the others.
At CorTec, we bring proven end-to-end expertise to address this complexity across every development stage. With more than a decade of experience in active implantable technologies, we are a fully integrated partner for neuromodulation medical device solutions, helping neurotech teams move from early concept to clinical deployment with fewer handoffs, less uncertainty, and faster progress.
Fragmentation Into Fast-Track: Protect Your Time And BudgetMost teams don’t fail because they lack ideas. They struggle when development becomes fragmented, when specialists, vendors, and workstreams all move in different directions.
CorTec is built to stop that. By unifying critical neurotech components and expertise under one integrated platform, we cut friction, reduce rework, and help you reach key milestones faster—saving you significant time and development spend.
A Complete Capability Stack, Brought Together Under One Roof
As a fully integrated partner, CorTec is set up to eliminate the gaps between disciplines. Our integrated capability stack covers the core building blocks of neuromodulation systems:
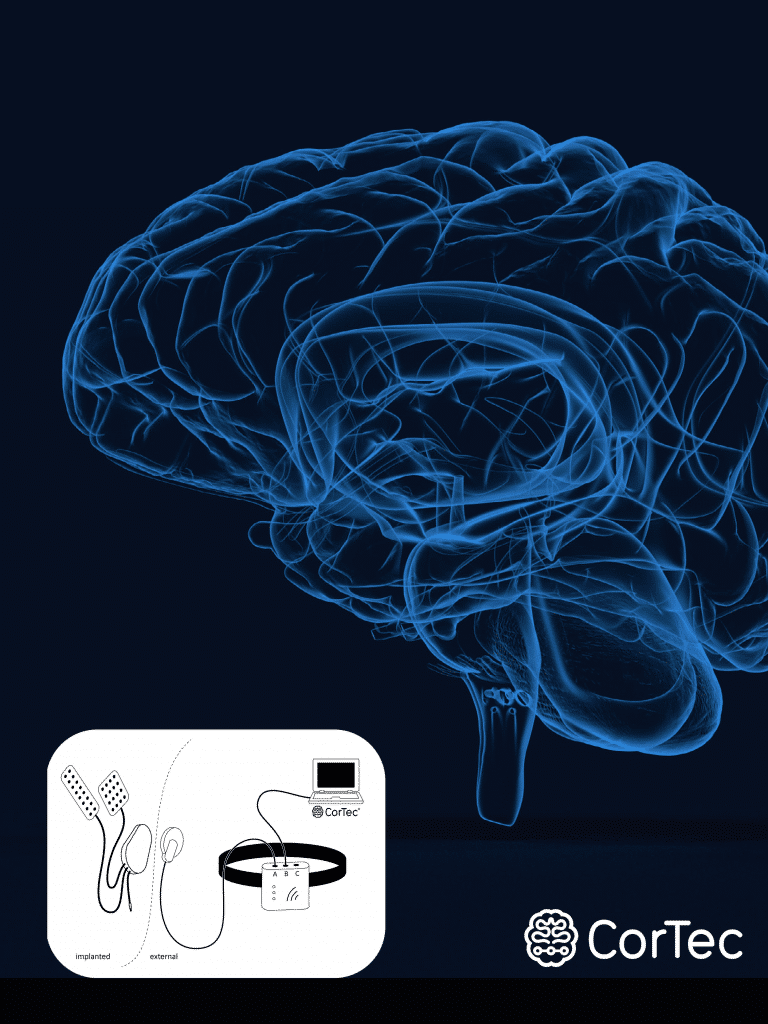
- Lead Technologies: From cuff and grid to paddle as well as DBS and percutaneous lead designs, our implantable leads support the full range of possibilities, engineered to your requirements with clinical-grade standards.
- Miniaturized Implant Packaging Reliable encapsulation is essential for long-term performance inside the body. Our high-channel packaging and precision silicone overmoulding enable stable, biocompatible device interfaces that are built for durability and consistency.
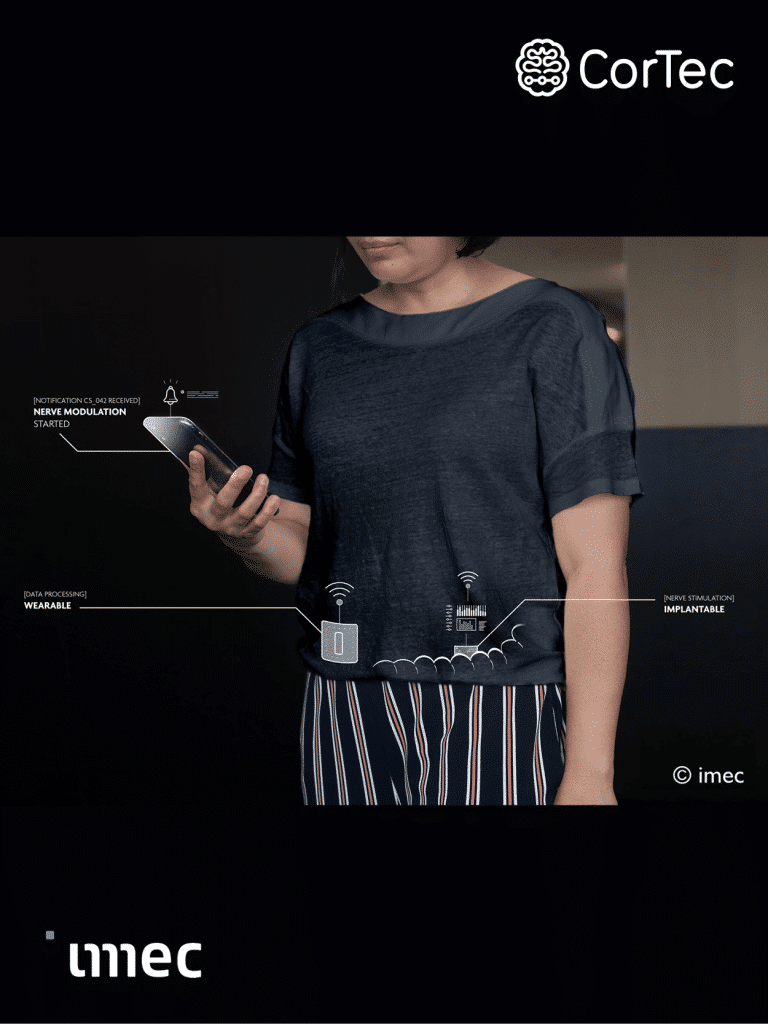
- Electronics & Software Development: A high-performance lead needs a high-performance system. CorTec develops embedded electronics, firmware, and clinical-grade software — from stimulation and sensing hardware to clinician-facing functionality.
What CorTec Covers For You
We support every stage of the device lifecycle:
- Concept to Prototype – turn clinical insights into robust engineering concepts and validated prototypes, fast.
- Testing & Regulatory – build in quality, biocompatibility, and documentation to clear approval hurdles with confidence.
- Manufacturing & Commercialization – scale reliable production and support a smooth ramp‑up and launch into the market.
- Designed for Manufacturing
CorTec is focused on neurotech innovators and established MedTech companies. In practice, that means bringing together deep technical, regulatory, and operational expertise with the execution discipline development teams need when timelines are tight—from early development through rampup and commercialization.
The result: integrated design thinking that helps de-risk decisions and improve outcomes across the full journey from surgical considerations to clinical translation and beyond.
At the Center of Medical Device’s Next Chapter
Cardiology defined the last three decades of device innovation. Neuromodulation is poised to define the next. Neuromodulation has moved well beyond its early use in epilepsy, depression, and Parkinson’s disease and is now rapidly expanding into new indications such as obstructive sleep apnea, autoimmune diseases, and diabetes. Demand for highprecision, miniaturized, fully implantable systems has never been higher, and the pace of innovation keeps accelerating. CorTec is positioned at the center of this transformation. As a fully integrated partner for neuromodulation medical device solutions, with the expertise, technology, and operational agility to help you move faster, with greater confidence, and with the shared goal of making the impossible possible.
Ready to Accelerate Your Neurotech Program?
Whether you are building your first implantable system or scaling into new indications, CorTec gives you the integrated capabilities, clinical-grade expertise, and structured execution needed to move with confidence.
Let’s build something that matters.